Explore Our Research
(Click on any research topic to learn more)
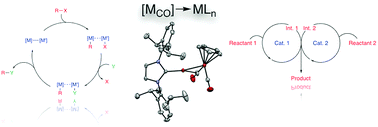
Heterobimetallic Reactivity
Replacing precious metals with earth-abundant alternatives is a key goal in developing more sustainable chemical technologies. Achieving this objective will require new design principles for reactive complexes built from abundant elements. One promising strategy is heterobimetallic cooperativity — harnessing the synergistic behavior of two distinct metal centers within a single complex to unlock reactivity that neither metal could achieve alone.
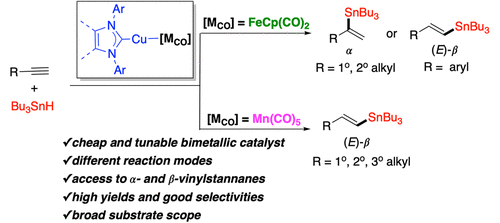
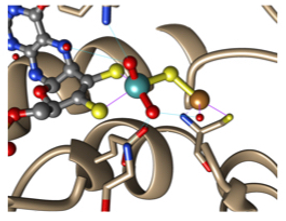
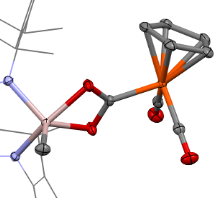
Our group has a longstanding interest in such systems. We have developed heterobimetallic complexes such as (NHC)Cu–FeCp(CO)₂ derivatives (NHC = N-heterocyclic carbene, Cp = cyclopentadienyl) that function as transition metal analogues of frustrated Lewis pairs, combining nucleophilic and electrophilic character in a single scaffold. Our work has explored their synthesis, electronic structure, reactivity, catalytic potential, and mechanisms.
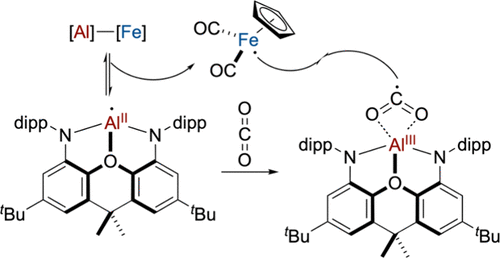
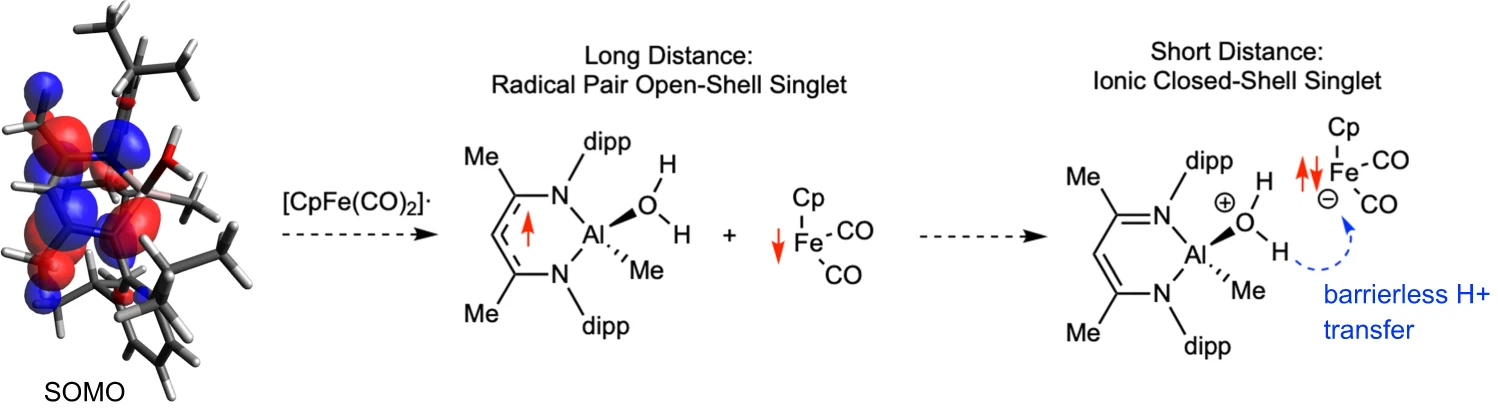
More recently, we have extended this concept to new LnAl–FeCp(CO)₂ complexes combining aluminum and iron. These systems act as frustrated radical pair analogues, enabling the generation of rare formally Al(II) species at ambient conditions. Studying these intermediates allows us to explore reactivity patterns that are unprecedented for aluminum chemistry. Ultimately, we aim to leverage this cooperative behavior to develop catalytic transformations based on aluminum and iron — the two most abundant metals in the Earth’s crust.
Currently, these projects are funded by U.S. Dept. of Energy grant DE-SC0021055.
Selected publications
Accepting new researchers:
Not accepting new researchers: