Explore Our Research
(Click on any research topic to learn more)
Electrolyte Design
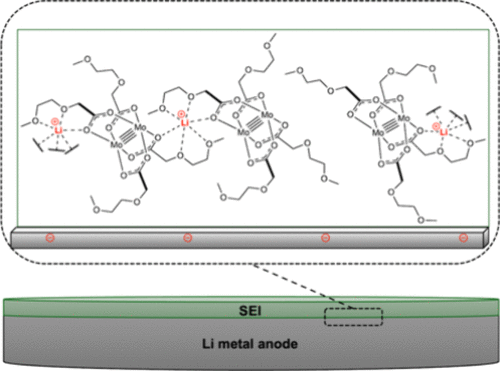
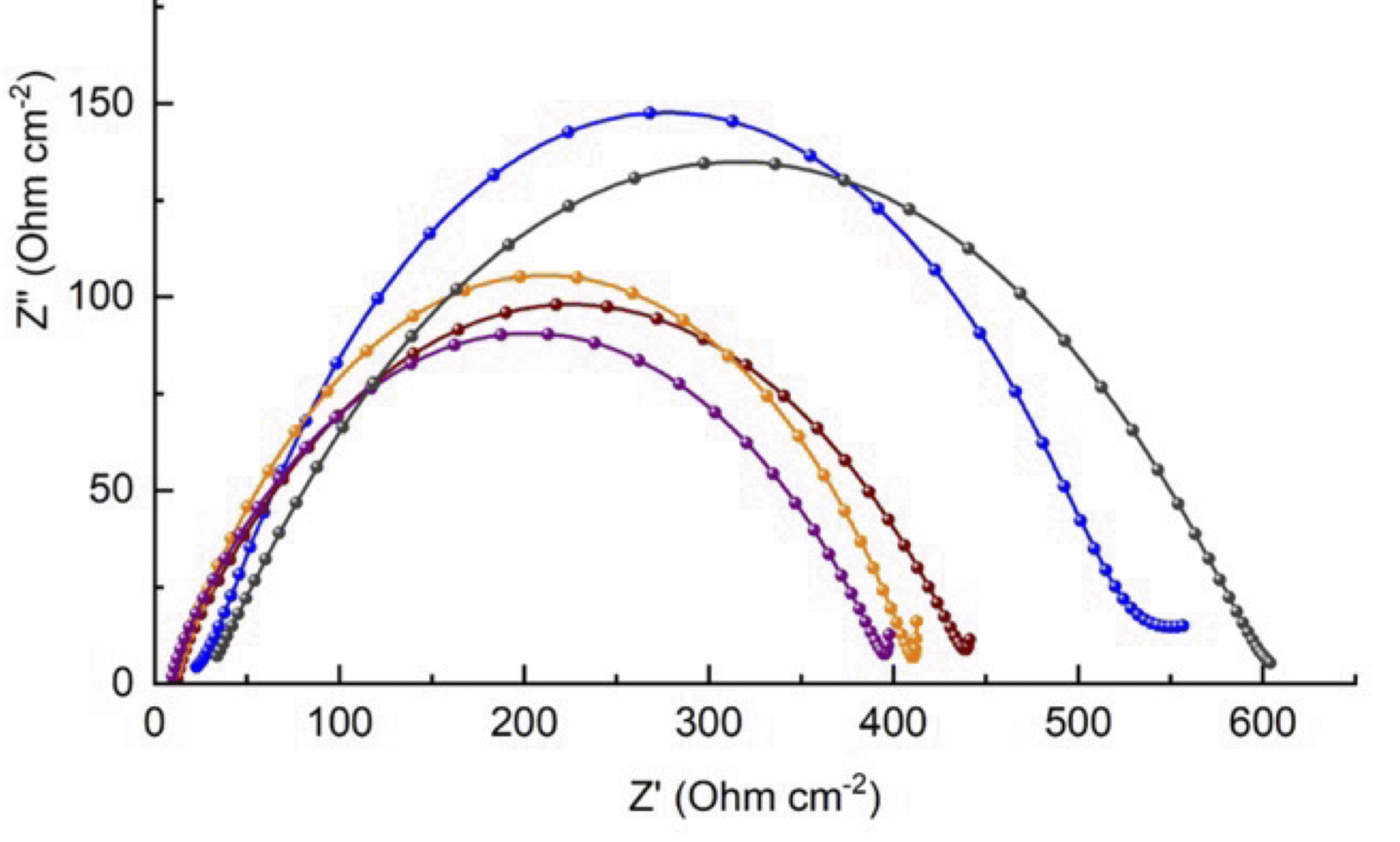
Emerging battery technologies based on metal anodes—including lithium, sodium, and magnesium—have the potential to deliver transformative advances in energy storage. These systems promise higher energy densities and lower costs than conventional lithium-ion batteries. However, their development is hindered by instability and safety risks stemming from uncontrolled reactions between the metallic anode and the battery electrolyte. A key challenge lies in engineering the solid–electrolyte interphase (SEI): the nanoscale region that forms where the anode meets the electrolyte. The composition and structure of the SEI critically affect both the performance and safety of next-generation batteries.
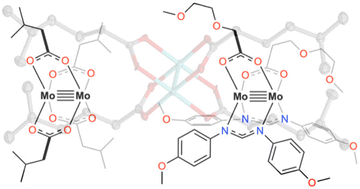
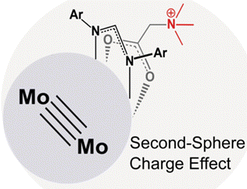
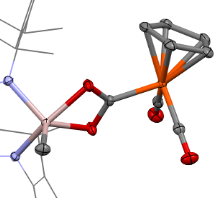
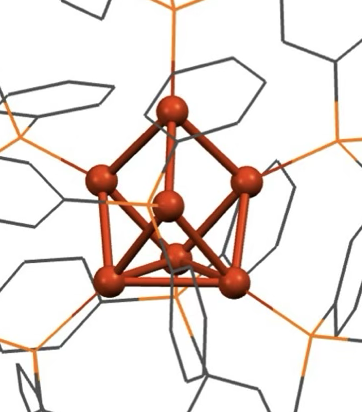
Our group serendipitously discovered a class of molybdenum-based coordination compounds that dramatically improve SEI properties on lithium metal anodes—even when used at millimolar concentrations. These compounds help stabilize the SEI, reducing reactivity while preserving rechargeability and electrochemical performance. We are now extending this chemistry to “beyond lithium” systems, such as sodium and magnesium, and investigating the mechanism of action through a combination of electrochemical studies and surface analysis. By leveraging molecular synthesis to systematically tune the structure of these electrolyte additives, we aim to build structure–function relationships that guide rational electrolyte design. This project combines our strengths in organometallic synthesis with electrochemistry and interfacial materials characterization, with the ultimate goal of enabling safer, more efficient energy storage technologies.
Currently, these projects are funded by NSF grant CHE-2350403.
Selected publications
Accepting new researchers:
Not accepting new researchers: